How Many Electrons In Lead
This is a SUPER easy guide on Lead element.
In fact, the table mentioned below is the perfect information box (Which gives y'all every unmarried detail about the Lead element in Periodic table.)
So if you want to know anything about Lead chemical element, then this guide is for you.
Let's swoop right into it!
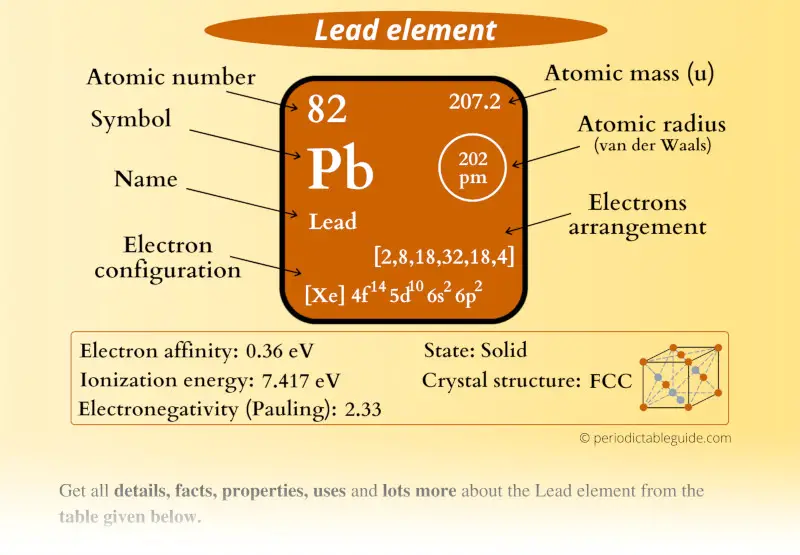
Pb Chemical element (Pb) Information
| Appearance of Lead |  Metallic gray advent |
| Country of Lead at STP | Solid |
| Position of Lead in Periodic table | 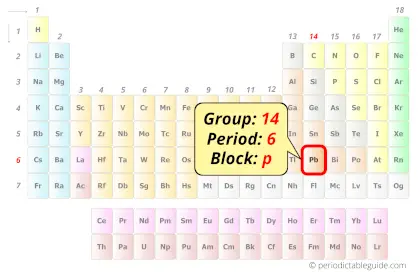 Group: fourteen, Period: half-dozen, Block:p |
| Category of Atomic number 82 element | 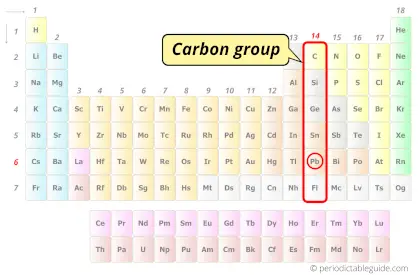 Carbon grouping |
| Atomic number of Lead, or Protons in Pb | 82 |
| Neutrons in Lead | 125 |
| Electrons in Lead | 82 |
| Symbol of Lead | Lead |
| Diminutive mass of Atomic number 82 |  207.2 u |
| Electrons arrangement in Lead or Bohr model of Lead | 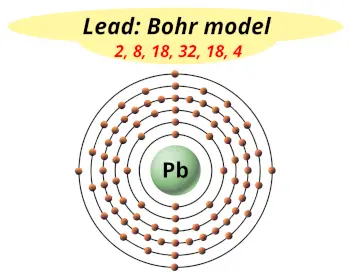 2, 8, xviii, 32, 18, 4 |
| Electronic configuration of Atomic number 82 | [Xe] 4f14 5d10 6s2 6pii |
| Atomic radius of Lead | 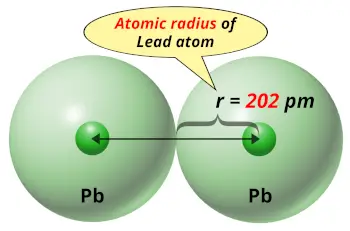 202 picometers (van der Waals radius) |
| Valence electrons in Lead | 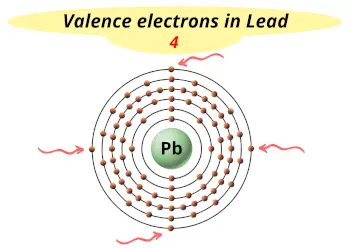 4 |
| 1st Ionization free energy of Pb | 7.417 eV |
| Electronegativity of Atomic number 82 | 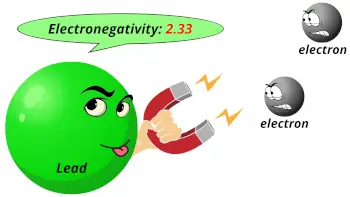 2.33 (Pauling calibration) |
| Crystal structure of Lead | 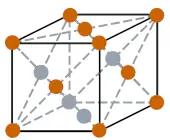 FCC (confront centered cubic) |
| Melting point of Atomic number 82 | 600.6 1000 or 327.4 °C or 621.4 °F |
| Boiling point of Lead | 2022 K or 1749 °C or 3180 °F |
| Density of Lead | eleven.34 g/cmiii |
| Main isotope of Lead | 208Pb |
| CAS number | 7439-92-1 |
Lead in Periodic tabular array
Lead element is in group fourteen and period 6 of the Periodic table. Lead is the p-block element and it belongs to carbon group.
←Motility to: Thallium (Tl) element – Periodic Table
→Move to: Bismuth (Bi) chemical element – Periodic Table
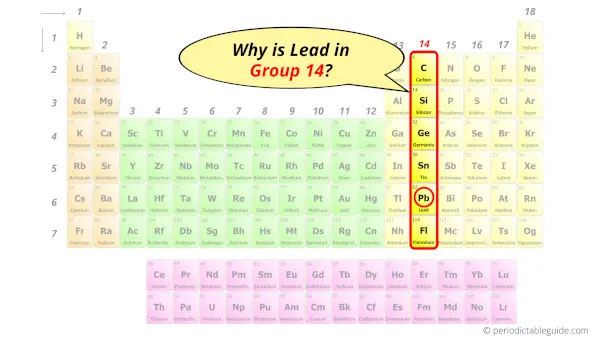
Why is Lead in Group xiv?
Practice you know, how many electrons can be accommodated in the first shell, 2nd trounce, tertiary shell, quaternary shell, etc…?
Here is the table showing the capacity of orbits to hold electrons.
Number of electrons in shells.
| Orbit / Crush (n) | Maximum no. of electrons this orbit can concur (two × northward two ) |
| K shell, north = 1 | 2 × 1² = ii |
| L beat, n = 2 | ii × 2² = viii |
| M trounce, n = iii | two × 3² = 18 |
| N trounce, n = 4 | 2 × 4² = 32 |
| . . . | . . . |
Thus,
- 1st shell can concur 2 electrons.
- 2nd beat out can concord 8 electrons.
- 3rd shell can hold 18 electrons.
- 4th shell tin can hold 32 electrons.
Now the atomic number of Lead (Lead) is 82.
Hence the Lead chemical element has electrons arrangement ii, eight, 18, 32, xviii, four.
This electron arrangement indicates that the outermost orbit of Atomic number 82 chemical element (Lead) has 4 electrons.
Hence, it lies in grouping 14.
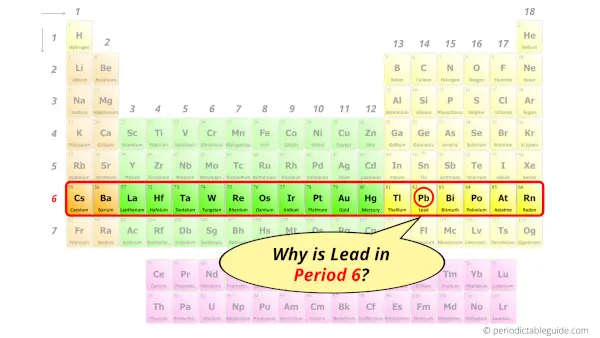
Why is Atomic number 82 in Period six?
Permit me enquire y'all a question.
How many shells does pb have?
It'southward 6. Right?
Yous have already seen the bohr model of lead atom in the above tabular array.
From the Bohr model, it can be found that the number of orbits or shells in lead is 6. Hence, every bit lead has 6 orbits, it lies in menstruum 6 of the Periodic table.
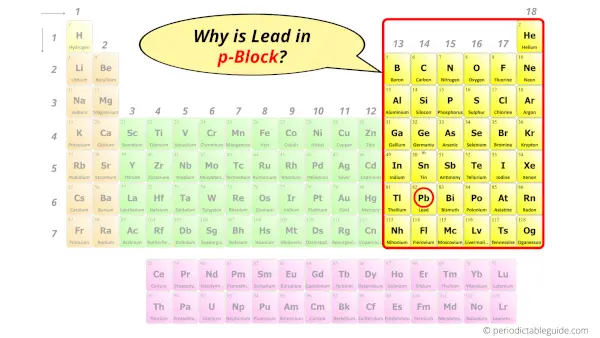
Why is Atomic number 82 in p-block?
Before knowing this reason, first of all I want to enquire you a unproblematic question.
How can you determine the blocks-wise position of elements?
The simple reply: The elements volition prevarication in the s, p, d or f block volition completely depend upon the subshell in which the terminal electron will enter.
For example; the electron configuration of lead is [Xe] 4ffourteen 5d10 6s2 6p2.
So the final electron of atomic number 82 enters the p-subshell or p-orbital.
Hence, lead is the p-block element.
v Interesting facts about Lead
Interesting facts almost lead element are mentioned below.
- The concentration of lead in the earth's crust is effectually 114 ppm past weight.
- Lead is primarily obtained from its ore lead sulfide (PbS). atomic number 82 is also obtained from the ores of copper, silverish and zinc.
- Pb is ane of the elements that was discovered since ancient times.
- Lead is a soft metal and it can be cut with a knife also.
- Lead is a poisonous element. As well much pb in the human trunk tin can crusade nervous problems equally well as information technology as well affects organs similar kidney, centre, intestines, etc.
Backdrop of Lead
The physical and chemical backdrop of lead element are mentioned below.
Concrete properties of Atomic number 82
Concrete properties of lead are mentioned beneath.
- Lead is a solid metallic at STP and information technology has a metallic grey appearance.
- Lead is a poor conductor of electricity.
- Lead is ductile as well as malleable metal. That ways it can exist fatigued into sparse wires and thin sheets.
- There are many isotopes of lead, simply out of those isotopes the isotope 208Pb is the most abundant (having affluence of approximately 52.iv%).
- Lead element has a lower melting point of 327.4 °C. And its boiling signal is 1749 °C.
- The crystal structure of pb is FCC (i.e face centered cubic).
Chemical properties of Atomic number 82
Chemical properties of lead are mentioned beneath.
- When the lead is freshly cut, its surface is bluish gray. Simply if it is kept open in air, and then information technology reacts with oxygen and starts tarnishing (which forms a grey oxide layer on it).
- The electron configuration of lead is [Xe] 4ffourteen 5dx 6s2 6p2, which shows that the last electron enters the p-orbital. Because of this reason, it is classified as a p-cake element on the periodic table.
- In the compound form, lead exists in its most common oxidation states +2 and +4. Apart from these oxidation states, other oxidation states likewise exist.
- The 1st ionization free energy of lead is 7.417 eV.
- The electronegativity of atomic number 82 is 2.33 on the Pauling scale.
Uses of Lead
Uses of atomic number 82 are mentioned beneath.
- Most of the lead produced nowadays is used in manufacturing atomic number 82 batteries.
- Lead is also used in solder materials as well as in covering materials for cables.
- In ancient times, lead was used in plumbing (as it is non corrosive). Merely due to its toxicity it is not used nowadays.
- Lead is also used in manufacturing radiation shields, bullets, etc.
- Lead acts as a superconductor at temperatures below seven.2 Yard. [1]
Costless Gift for y'all: Interactive Periodic Table
Let me tell you how this Interactive Periodic Table will assist you in your studies.
1). You lot can effortlessly discover every single detail well-nigh the elements from this single Interactive Periodic table.
ii). You will get the detailed information about the periodic table which will convert a newbie into pro.
3). You will also get the HD images of the Periodic table (for FREE).
Checkout Interactive Periodic table and download it's high resolution epitome at present (Information technology'southward FREE)
References:
Chemical information: Wikipedia, Chemspider
Pb chemical element: Image by Alchemist-hp (www.pse-mendelejew.de), FAL, via Wikimedia Eatables
How Many Electrons In Lead,
Source: https://periodictableguide.com/lead-pb-element-periodic-table/
Posted by: bellwased1993.blogspot.com


0 Response to "How Many Electrons In Lead"
Post a Comment